LA PILE A COMBUSTIBLE
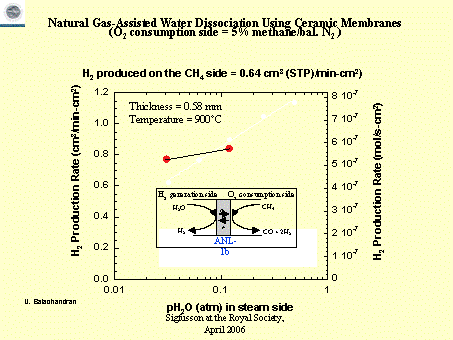
Use of mixed conducting membranes to produce hydrogen
by water dissociation
U. Balachandran , , T. H. Lee, S. Wang and S. E. Dorris
Energy Technology Division, Argonne National Laboratory,
9700 South Cass Avenue, Argonne, IL 60439, USA
International Journal of Hydrogen Energy
Volume 29, Issue 3 , March 2004, Pages 291