http://www.technologyreview.com/
Researchers have found a cheap and easy way
to store the energy made by solar power.

Splitting water:
Daniel Nocera poses with a device for breaking down water into hydrogen
and oxygen. The device uses an inexpensive catalyst that he has developed.
Researchers have made
a major advance in inorganic chemistry that could lead to a cheap way to
store energy from the sun. In so doing, they have solved one of the key
problems in making solar energy a dominant source of electricity.
Daniel
Nocera, a professor of chemistry at MIT, has developed a catalyst that
can generate oxygen from a glass of water by splitting water molecules.
The reaction frees hydrogen ions to make hydrogen gas. The catalyst, which
is easy and cheap to make, could be used to generate vast amounts of hydrogen
using sunlight to power the reactions. The hydrogen can then be burned
or run through a fuel cell to generate electricity whenever it's needed,
including when the sun isn't shining.
Solar power is ultimately
limited by the fact that the solar cells only produce their peak output
for a few hours each day. The proposed solution of using sunlight to split
water, storing solar energy in the form of hydrogen, hasn't been practical
because the reaction required too much energy, and suitable catalysts were
too expensive or used extremely rare materials. Nocera's catalyst clears
the way for cheap and abundant water-splitting technologies.
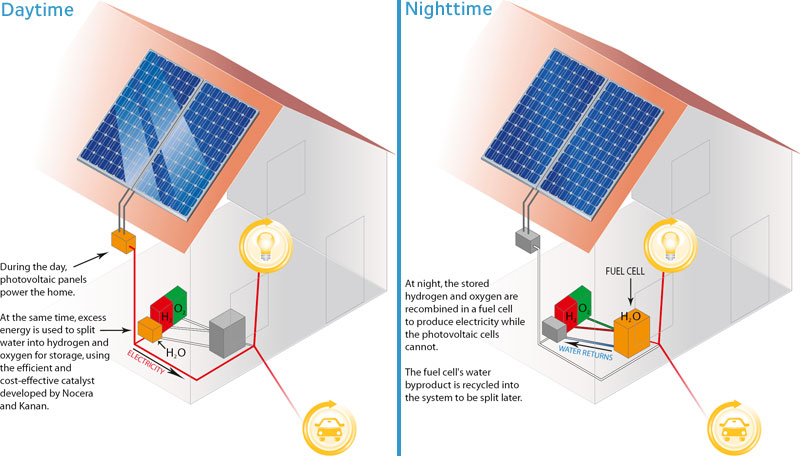
HOW IT WORKS
Illustration of Dr. Nocera's "artificial photosynthesis" system.
Nocera's advance represents
a key discovery in an effort by many chemical research groups to create
artificial
photosynthesis--mimicking how plants use sunlight to split water to
make usable energy. "This discovery is simply groundbreaking," says
Karsten
Meyer, a professor of chemistry at Friedrich Alexander University,
in Germany. "Nocera has probably put a lot of researchers out of business."
For solar power, Meyer says, "this is probably the most important single
discovery of the century."
The new catalyst marks
a radical departure from earlier attempts. Researchers, including Nocera,
have tried to design molecular catalysts in which the location of each
atom is precisely known and the catalyst is made to last as long as possible.
The new catalyst, however, is amorphous--it doesn't have a regular structure--and
it's relatively unstable, breaking down as it does its work. But the catalyst
is able to constantly repair itself, so it can continue working.
In his experimental
system, Nocera immerses an indium tin oxide electrode in water mixed with
cobalt and potassium phosphate. He applies a voltage to the electrode,
and cobalt, potassium, and phosphate accumulate on the electrode, forming
the catalyst. The catalyst oxidizes the water to form oxygen gas and free
hydrogen ions. At another electrode, this one coated with a platinum catalyst,
hydrogen ions form hydrogen gas. As it works, the cobalt-based catalyst
breaks down, but cobalt and potassium phosphate in the solution soon re-form
on the electrode, repairing the catalyst.
Nocera created the catalyst
as part of a research program whose goal was to develop artificial photosynthesis
that works more efficiently than photosynthesis and produces useful fuels,
such as hydrogen. Nocera has solved one of the most challenging parts of
artificial photosynthesis: generating oxygen from water. Two more steps
remain. One is replacing the expensive platinum catalyst for making hydrogen
from hydrogen ions with a catalyst based on a cheap and abundant metal,
as Nocera has done with the oxygen catalyst.
Finding a cheaper catalyst
for making hydrogen shouldn't be too difficult, says John
Turner,
a principal investigator at the National Renewable Energy Laboratory,
in Golden, CO. Indeed, Nocera says that he has promising new materials
that might work, and other researchers also have likely candidates. The
second remaining step in artificial photosynthesis
ihttp://www.sciam.com/article.cfm?id=hydrogen-power-on-the-cheap&sc=WR_20080805
http://www.sciam.com/article.cfm?id=hydrogen-power-on-the-cheap&sc=WR_20080805s
developing a material that absorbs sunlight, generating the electrons needed
to power the water-splitting catalysts. That will allow Nocera's catalyst
to run directly on sunlight; right now, it runs on electricity taken from
an outlet.
There's also still much
engineering work to be done before Nocera's catalyst is incorporated into
commercial devices. It will, for example, be necessary to improve the rate
at which his catalyst produces oxygen. Nocera and others are confident
that the engineering can be done quickly because the catalyst is easy to
make, allowing a lot of researchers to start working with it without delay.
"The beauty of this system is, it's so simple that many people can immediately
jump on it and make it better," says Thomas
Moore, a professor of chemistry and biochemistry at Arizona State University. |
LE
MONDE | 08.08.08
C'est un rêve d'écologiste:
produire de l'énergie avec du soleil et de l'eau, à la manière
de la photosynthèse réalisée par les plantes. Ce rêve
pourrait devenir réalité, si l'on en croit les travaux, publiés
dans la revue Science du 1er août, de deux chimistes américains
du Massachusetts Institute of Technology (MIT), Daniel Nocera et Matthew
Kanan. Dans la nature, les végétaux utilisent la lumière
comme source d'énergie pour fabriquer du sucre à partir de
gaz carbonique et d'eau, dont les molécules sont décomposées
entre d'un côté l'oxygène et de l'autre l'hydrogène.
C'est cette réaction qu'imite l'électrolyse de l'eau, consistant
à dissocier les molécules liquides en oxygène et hydrogène
gazeux, à l'aide d'un courant électrique circulant entre
deux électrodes. Un procédé connu de tous les écoliers
et maîtrisé de longue date, puisque la première électrolyse
de l'eau a été effectuée en 1800 par deux chimistes
britanniques.
L'usage à grande
échelle de cette technique est toutefois limité par des obstacles
économiques. Les installations industrielles d'électrolyse
de l'eau sont en effet complexes et coûteuses, notamment parce qu'elles
nécessitent des catalyseurs (activant les réactions) qui
sont habituellement faits de platine, métal cher. L'idée
des chercheurs du MIT est d'exploiter, pour provoquer l'électrolyse,
le rayonnement solaire converti en électricité par les cellules
photovoltaïques de panneaux équipant les maisons et les bâtiments
publics. Et de recourir à des matériaux moins onéreux.
PILE À COMBUSTIBLE
Daniel Nocera et Matthew
Kanan ont montré que la dissociation de l'eau en oxygène
et hydrogène pouvait être réalisée avec une
électrode en oxyde d'indium (métal proche de l'aluminium
que l'on trouve en petites quantités dans les minerais de zinc)
dopé à l'étain, placée dans un bain d'eau additionnée
de cobalt et de phosphate de potassium. Lesquels s'avèrent, en présence
d'un courant électrique, des catalyseurs efficaces.
Tout l'intérêt
de l'opération est d'obtenir, in situ et à moindre coût,
de l'hydrogène. Celui-ci pourra ensuite être recombiné
à de l'oxygène pour produire de l'électricité,
selon le procédé inverse de l'électrolyse mis en oeuvre
dans les piles à combustible. Dans la pratique, les auteurs imaginent
des habitations dotées de capteurs photovoltaïques qui, pendant
les heures d'ensoleillement, les alimenteraient en électricité.
L'excès d'électricité servirait à produire
de l'hydrogène qui, la nuit, serait recombiné à de
l'oxygène dans une pile à combustible. L'énergie nécessaire
au foyer - voire à une voiture électrique - serait ainsi
fournie en permanence et à demeure.
Ces résultats
sont jugés "très intéressants" par Paul Lucchese,
directeur du programme "Nouvelles technologies de l'énergie" au
Commissariat à l'énergie atomique (CEA), qui travaille également
sur la filière hydrogène. Ils constituent "une brique
supplémentaire dans un ensemble de recherches menées depuis
quelques années sur les systèmes biomimétiques, s'inspirant
de la photosynthèse naturelle pour produire de l'hydrogène".
Toutefois, souligne-t-il, "il ne s'agit encore que d'une expérience
de laboratoire et il reste un énorme travail technologique à
accomplir avant de disposer de systèmes fonctionnels".
Pierre Le Hir
Published Online July 31, 2008
Science DOI: 10.1126/science.1162018
Science
Express Index
Reports
Submitted on June 19, 2008
Accepted on July 18, 2008
In Situ Formation of an Oxygen-Evolving Catalyst
in Neutral Water Containing Phosphate and CO2+
Matthew W. Kanan 1 and Daniel G. Nocera
1* 1 Department of Chemistry, 6-335, Massachusetts Institute of Technology,
Cambridge, MA 02139–4307, USA.
* To whom correspondence should be addressed.
Daniel G. Nocera , E-mail: nocera@mit.edu
The utilization of solar
energy on a large scale requires its storage. In natural photosynthesis,
energy from sunlight is used to rearrange the bonds of water to O2
and H2-equivalents. The realization of artificial systems that
perform similar "water splitting" requires catalysts that produce O2
from water without the need for excessive driving potentials. Here, we
report such a catalyst that forms upon the oxidative polarization of an
inert indium tin oxide electrode in phosphate-buffered water containing
CO2+. A variety of analytical techniques indicates
the presence of phosphate in an approximate 1:2 ratio with cobalt in this
material. The pH dependence of the catalytic activity also implicates HPO42–
as the proton acceptor in the O2-producing reaction. This catalyst
not only forms in situ from earth-abundant materials but also operates
in neutral water under ambient conditions. |


