LE FUTUR DU SOLAIRE PV
Nouveautés internationales
2007
| L'équipe de Charles Lieber
à Harvard University (Cambridge, MA) a mis au point une cellule
solaire constituée d'un nanofil de silicium de 300 nanomètres
de diamètre formé de trois couches concentriques déposées
par méthode VLS (Vapeur Liquide Solide): le coeur est dopé
P, la couche intermédiaire est intrinsèque et la couche extérieure
est dopée N, l'ensemble formant une jonction PIN radiale. Ce type
de structure coaxiale permet d'augmenter sensiblement l'efficacité
de collection des porteurs ainsi que le rendement global par rapport à
des structures conventionnelles utilisant le même semiconducteur
monocristallin.
Les résultats obtenus montrent que ce type de cellule solaire à base de nanofil peut fournir une puissance électrique pouvant atteindre 200 pW et présenter un rendement de conversion stable de 3,4%. Cette valeur peut paraître faible comparée à celles obtenues avec les cellules solaires à base de silicium monocristallin, mais cette approche innovante est attractive car elle devrait permettre de réaliser des dispositifs électroniques auto-alimentés grâce à l'implantation sur le circuit même d'éléments photovoltaïques constitués de nanofils empilés. |
Sources:
- http://www.nanowerk.com/news/newsid=2987.php - Publication parue dans Nature: "Coaxial silicon nanowires as solar cells and nanoelectronic power sources" - Bozhi Tian1, Xiaolin Zheng1, Thomas J. Kempa1, Ying Fang1, Nanfang Yu, Guihua Yu, Jinlin Huang & Charles M. Lieber - Nature Vol 449|, 18/10/2007 - http://cmliris.harvard.edu/publications/2000s/2007/nature06181.pdf Rédacteur: Romaric Fayol - deputy-phys.mst@consulfrance-houston.org Origine: BE Etats-Unis numéro 97 (29/10/2007) - Ambassade de France aux Etats-Unis / ADIT |

| Ceci n'est pas une feuille de
papier ou de plastique noir, mais bien une pile électrique. Et la
dernière trouvaille des chercheurs du Rensselaer Polytechnic Institute
(RPI) américain. Grâce à la nanotechnologie, ils sont
parvenus à produire une structure moléculaire très
proche du papier, avec 90% de cellulose. Les 10% restant sont formés
de nanotubes de carbone, qui agissent comme des électrodes, et peuvent
conduire du courant.
Résultat: une batterie qui fonctionne comme une pile au lithium, et qui est aussi capable de stocker de l'électricité comme un super-condensateur. Avec d'autres avantages: comme une feuille de papier, on peut la plier, la rouler, et même la découper où la déchirer en morceaux. Chaque élément séparé conservera ses propriétés de stockage d'électricité. On peut également imprimer textes et dessins sur la pile. Autre innovation: la pile, qui utilise comme électrolyte (milieu conducteur) une solution ionisée (salée), peut voir ses propriétés améliorées lorsqu'on y ajoute de la sueur ou du sang humains. Mieux: lorsqu'on empile plusieurs feuilles, les capacités de stockage de la pile augmentent. Elle peut fonctionner normalement par des températures allant de -37,8 degrés à 148,9 degrés. Et elle est enfin infiniment moins nocive pour l'environnement que les piles au lithium. Les applications de cette innovation sont multiples, par exemple dans l'électronique, où l'on recherche toujours la miniaturisation, mais aussi dans le domaine médical. Reste à développer un processus de production industriel, ce à quoi s'emploie le RPI. Beyond batteries: Storing power in a sheet of paper Researchers turn everyday paper into resilient,
rechargeable energy storage device
|
"We're not putting pieces together it's
a single, integrated device," he said. "The components are molecularly
attached to each other: the carbon nanotube print is embedded in the paper,
and the electrolyte is soaked into the paper. The end result is a device
that looks, feels, and weighs the same as paper."
The creation of this unique nanocomposite paper drew from a diverse pool of disciplines, requiring expertise in materials science, energy storage, and chemistry. Along with Linhardt, authors of the paper include Pulickel M. Ajayan, professor of materials science and engineering, and Omkaram Nalamasu, professor of chemistry with a joint appointment in materials science and engineering. Senior research specialist Victor Pushparaj, along with postdoctoral research associates Shaijumon M. Manikoth, Ashavani Kumar, and Saravanababu Murugesan, were co-authors and lead researchers of the project. Other co-authors include research associate Lijie Ci and Rensselaer Nanotechnology Center Laboratory Manager Robert Vajtai. The researchers used ionic liquid, essentially a liquid salt, as the battery's electrolyte. It's important to note that ionic liquid contains no water, which means there's nothing in the batteries to freeze or evaporate. "This lack of water allows the paper energy storage devices to withstand extreme temperatures," Kumar said. Along with use in small handheld electronics, the paper batteries' light weight could make them ideal for use in automobiles, aircraft, and even boats. The paper also could be molded into different shapes, such as a car door, which would enable important new engineering innovations. "Plus, because of the high paper content and lack of toxic chemicals, it's environmentally safe," Shaijumon said. Paper is also extremely biocompatible and these new hybrid battery/supercapcitors have potential as power supplies for devices implanted in the body. The team printed paper batteries without adding any electrolytes, and demonstrated that naturally occurring electrolytes in human sweat, blood, and urine can be used to activate the battery device. "It's a way to power a small device such as a pacemaker without introducing any harsh chemicals such as the kind that are typically found in batteries into the body," Pushparaj said. The materials required to create the paper batteries are inexpensive, Murugesan said, but the team has not yet developed a way to inexpensively mass produce the devices. The end goal is to print the paper using a roll-to-roll system similar to how newspapers are printed. "When we get this technology down, we'll basically have the ability to print batteries and print supercapacitors," Ajayan said. "We see this as a technology that's just right for the current energy market, as well as the electronics industry, which is always looking for smaller, lighter power sources. Our device could make its way into any number of different applications." The team of researchers has already filed a patent protecting the invention. They are now working on ways to boost the efficiency of the batteries and supercapacitors, and investigating different manufacturing techniques. "Energy storage is an area that can be addressed by nanomanufacturing technologies and our truly inter-disciplinary collaborative activity that brings together advances and expertise in nanotechnology, room-temperature ionic liquids, and energy storage devices in a creative way to devise novel battery and supercapacitor devices," Nalamasu said. Contact: Michael Mullaney
|
| Le prix moyen de l'installation d'un
système solaire dans une maison australienne de taille moyenne est
d'environ 20.000 dollars australiens. On estime que le coût élevé
de la production des constituants en silicium des cellules photovoltaïques
est responsable de 45% du prix de revient du système installé.
Les cellules en couches minces représentent l'option la moins chère mais leur rendement de conversion reste faible (8 à 10%) par rapport à celui des cellules à base de silicium cristallin de type "wafer" (24,5%) qui sont beaucoup plus chères. L'équipe Photovoltaics Special Research Center de l'université de Nouvelle-Galles du Sud, à Sydney, qui détient le record mondial du taux de conversion (24,5%) pour une cellule photovoltaïque au silicium cristallin, a amélioré notablement les taux de conversion de cellules en couches minces. |
Ils ont réussi à augmenter d'un facteur
16 la capacité d'absorption de la lumière de couches minces
de 1,25 micro-m d'épaisseur, pour une lumière de longueur
d'onde de 1050 nm. Ils auraient également multiplié par 7
le coefficient d'absorption de la lumière (1200 nm) de cellules
de type "wafer". Cette augmentation du taux d'absorption se traduit par
un rendement plus élevé, compris entre 13 et 15%. Cette amélioration
est significative car le silicium absorbe faiblement la lumière
dans cette partie du spectre.
La méthode utilisée consiste à déposer un film d'argent d'environ 10 micro-m d'épaisseur sur la surface de la cellule solaire et de la chauffer ensuite à 200°C. Sous l'effet de la chaleur, le film se craquelle en domaines d'environ 100 nm de largeur. Les nanoparticules d'argent excitent des plasmons de surface qui augmentent l'absorption de la lumière. |
| Escape route:
Electrons created in a nanoparticle-based solar cell have to follow a circuitous path (red line) to reach an electrode. Many don't make it, lowering the efficiency of these cells. Researchers at Notre Dame have used carbon nanotubes to help the electrons reach the electrode, improving efficiency: 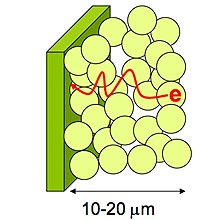 By Kevin Bullis Researchers at University of Notre Dame, in Indiana, have demonstrated a way to significantly improve the efficiency of solar cells made using low-cost, readily available materials, including a chemical commonly used in paints. The researchers added single-walled carbon nanotubes to a film made of titanium-dioxide nanoparticles, doubling the efficiency of converting ultraviolet light into electrons when compared with the performance of the nanoparticles alone. The solar cells could be used to make hydrogen for fuel cells directly from water or for producing electricity. Titanium oxide is a main ingredient in white paint. |
The approach, developed by Notre Dame professor
of chemistry and biochemistry Prashant Kamat and his colleagues, addresses
one of the most significant limitations of solar cells based on nanoparticles.
(See "Silicon
and Sun.") Such cells are appealing because nanoparticles have a great
potential for absorbing light and generating electrons. But so far, the
efficiency of actual devices made of such nanoparticles has been considerably
lower than that of conventional silicon solar cells. That's largely because
it has proved difficult to harness the electrons that are generated to
create a current.
Indeed, without the carbon nanotubes, electrons generated when light is absorbed by titanium-oxide particles have to jump from particle to particle to reach an electrode. Many never make it out to generate an electrical current. The carbon nanotubes "collect" the electrons and provide a more direct route to the electrode, improving the efficiency of the solar cells. As they wrote online in the journal Nano Letters, the Notre Dame researchers form a mat of carbon nanotubes on an electrode. The nanotubes serve as a scaffold on which the titanium-oxide particles are deposited. "This is a very simple approach for bringing order into a disordered structure," Kamat says. The new carbon-nanotube and nanoparticle system is not yet a practical solar cell. That's because titanium oxide only absorbs ultraviolet light; most of the visible spectrum of light is reflected rather than absorbed. But researchers have already demonstrated ways to modify the nanoparticles to absorb the visible spectrum. In one strategy, a one-molecule-thick layer of light-absorbing dye is applied to the titanium-dioxide nanoparticles. Another approach, which has been demonstrated experimentally by Kamat, is to coat the nanoparticles with quantum dots--tiny semiconductor crystals. Unlike conventional materials in which one photon generates just one electron, quantum dots have the potential to convert high-energy photons into multiple electrons. Several other groups are exploring approaches to improve the collection of electrons within a cell, including forming titanium-oxide nanotubes or complex branching structures made of various semiconductors. But experts say that Kamat's work could be a significant step in creating cheaper, more-efficient solar cells. "This is very important work," says Gerald Meyer, professor of chemistry at Johns Hopkins University. "Using carbon nanotubes as a conduit for electrons from titanium oxide is a novel idea, and this is a beautiful proof-of-principle experiment." |